Kayla M. Kwon
Abstract
Glioblastoma, also known as glioblastoma multiforme, is a highly aggressive and malignant brain tumor that arises from glial cells that accumulates on the brain or spine (Medscape, n.d.). We will analyze the clinical applications used to diagnose glioblastoma and the costs of treatment (Medscape, n.d.). Glioblastoma is currently a molecular diagnosis; genetic studies for IDH and H3 status are essential, and genetic studies for TERT promoter mutation, EGFR gene amplification, and +7/–10 chromosome copy number changes are also important (Medscape, n.d.). Furthermore, excluding a metabolic or infectious process with routine laboratory studies is critical in the evaluation of an otherwise healthy patient who presents with new-onset seizures or neurologic deficit (Medscape, n.d.). Researchers are working to identify a biomarker that could be used to diagnose glioblastoma with a noninvasive liquid biopsy taken from blood or cerebrospinal fluid, but this technology has not been fully realized (Medscape, n.d.).
Introduction
This paper begins by outlining the methodology used throughout the review. It then examines the mechanisms used to diagnose glioblastoma, with emphasis on the commonly used imaging modality in hospitals today (Radiological Society of North America & American College of Radiology [RSNA & ACR], n.d.-a). The risks and limitations associated with these diagnostic mechanisms are discussed, followed by an exploration of a representative study designed to establish a definitive diagnosis of glioblastoma, characterize its molecular profile, and exclude alternative etiologies in patients presenting with new neurological symptoms (Medscape, n.d.).
There are also similar conditions to glioblastoma such as anaplastic astrocytoma, which also originates from astrocytes (same cell lineage) and can look very similar to glioblastoma on MRIs (Medscape, n.d.). They share similar symptoms, including seizures and neurological deficits (Medscape, n.d.). However, a key difference is that anaplastic astrocytomas are less aggressive than glioblastoma (Grade 4) and typically show lower levels of necrosis and less contrast enhancement (Medscape, n.d.). Anaplastic astrocytoma is often considered a precursor that can progress into glioblastoma, making early identification clinically important (Medscape, n.d.). This literature review investigates what the neural and molecular indicators of glioblastoma are.
Methodology
This is a literature review (secondary data analysis) that examines real-world research on glioblastoma. It focuses on the diagnostic mechanisms and devices used to evaluate the disease and explores how glioblastoma affects patients’ daily lives and clinical care (Medscape, n.d.). However, there are limitations on studies conducted on glioblastoma due to its aggressive nature and poor prognosis (Medscape, n.d.). The core evidence relies on a specific study: Medscape’s Glioblastoma Workup (Medscape, n.d.).
Mechanisms for Diagnosis
There are a variety of mechanisms used to diagnose glioblastoma within patients. However, the primary imaging modality used to identify glioblastoma is magnetic resonance imaging with contrast (MRI) due to its superior soft-tissue visualization (RSNA & ACR, n.d.-a).
Magnetic Resonance Imaging with Contrast (MRI)
Magnetic resonance imaging is a mechanism that uses a strong magnetic field and radio waves to create detailed images of the body (National Institute of Biomedical Imaging and Bioengineering [NIBIB], n.d.). MRIs measure signals from hydrogen atoms, mostly in water and fat, within body tissues (NIBIB, n.d.). This produces very clear images of soft tissues, including organs such as the brain (RSNA & ACR, n.d.-a).
MRIs with contrast involve the use of contrast agents, which are substances injected into the bloodstream before or during a scan (RSNA & ACR, n.d.-b). The most common substance used during these scans is gadolinium (U.S. Food & Drug Administration [FDA], 2023). Gadolinium changes how nearby hydrogen atoms respond to the MRI’s magnetic field, making certain areas appear brighter or more visible on scans (FDA, 2023). MRIs with contrast are the most common mechanism used to identify and diagnose glioblastoma in patients (RSNA & ACR, n.d.-a).
Implementation of Magnetic Resonance Imaging (MRI) with Contrast
The process of administering an MRI with contrast begins when the patient lies on the scanner table. A safe dose of gadolinium is injected into the bloodstream through an intravenous (IV) line (National Jewish Health, n.d.). An MRI sequence is then run, and radiologists examine the images for bright, enhanced areas that may indicate tumor presence or abnormal tissue (Mass General Hospital, n.d.). This process does not involve radiation, unlike CT scans, and typically takes between 30 and 60 minutes (RSNA & ACR, n.d.-a; National Jewish Health, n.d.).
Risks and Limitations
Like all diagnostic mechanisms, MRI with contrast carries some risks. While gadolinium is generally safe, it may cause mild side effects, including nausea, headache, or a cold sensation at the injection site (RSNA & ACR, n.d.-b). Rarely, individuals with severe kidney disease should not receive gadolinium-based contrast agents (FDA, 2023). At extremely high doses, gadolinium toxicity can be fatal, although such occurrences are rare in clinical settings (FDA, 2023). Finally, MRI alone cannot definitively diagnose cancer, which is why a biopsy is required for confirmation of glioblastoma (Medscape, n.d.).
Clinical Presentation & Diagnosis
Symptoms of glioblastoma vary depending on tumor location and size. Common symptoms include headaches, seizures, neurological deficits, and increased intracranial pressure (Medscape, n.d.).
Tables
Table 1
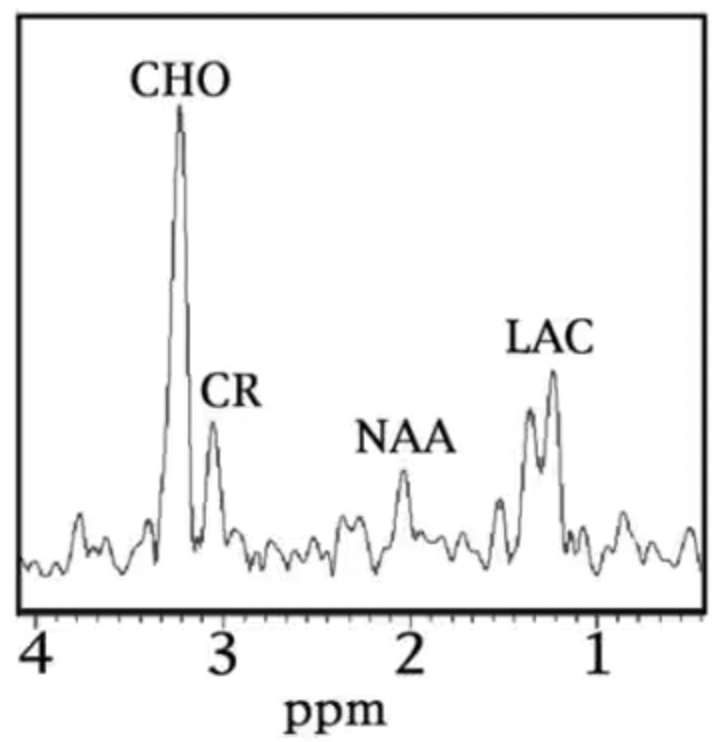
This magnetic resonance (MR) spectroscopy spectrum is from brain tissue in a patient with glioblastoma (GBM) (Medscape, n.d.). MR spectroscopy does not show anatomy like a standard MRI; instead, it displays the chemical composition of brain tissue (Medscape, n.d.). The x-axis (ppm) represents chemical shift, while the y-axis depicts metabolite concentration (Medscape, n.d.).
A characteristic neoplastic spectrum is present, marked by an increased choline (CHO) peak, decreased N-acetylaspartate (NAA) peak, and elevated lactate (LAC) peak (Medscape, n.d.). High choline reflects increased cell membrane turnover from rapid tumor cell growth, while reduced NAA indicates loss or replacement of normal neurons by tumor tissue (Medscape, n.d.). Lactate elevation signifies anaerobic metabolism due to tumor hypoxia and necrosis, which is common in high-grade tumors such as glioblastoma (Medscape, n.d.). Creatine (CR) is often reduced or unchanged, resulting in elevated CHO:CR and CHO:NAA ratios (Medscape, n.d.). This metabolic pattern helps distinguish glioblastoma from radiation necrosis, hemorrhage, or non-tumor injury and has prognostic value, as elevated choline/NAA ratios are associated with poorer outcomes (Medscape, n.d.).
Conclusion
The neural and molecular indicators of glioblastoma are reflected in characteristic changes in brain metabolism and tissue composition. Molecularly, glioblastoma demonstrates increased choline, decreased N-acetylaspartate, and elevated lactate levels, indicating rapid tumor growth, neuronal loss, and hypoxic metabolism (Medscape, n.d.). Neurally, these metabolic changes correspond to infiltration and destruction of normal brain tissue, hypoxia, and necrosis, highlighting the tumor’s aggressive nature (Medscape, n.d.). Clinically, MRI with contrast plays a critical role in identifying glioblastoma, although biopsy is required for definitive diagnosis (RSNA & ACR, n.d.-a; Medscape, n.d.). Despite advancements in diagnostic imaging, limitations remain due to the scarcity of long-term studies. Future research may focus on rare forms such as spinal glioblastoma and the development of noninvasive diagnostic biomarkers (Medscape, n.d.).
References
Mass General Hospital. (n.d.). MRI contrast: How it works and safety. https://www.massgeneral.org/imaging/programs-and-services/mri/mri-contrast
Medscape. (n.d.). Glioblastoma workup. https://emedicine.medscape.com/article/283252-workup
National Institute of Biomedical Imaging and Bioengineering. (n.d.). Magnetic resonance imaging (MRI). https://www.nibib.nih.gov/science-education/science-topics/magnetic-resonance-imaging-mri
National Jewish Health. (n.d.). MRI with intravenous contrast: Information for patients. https://www.nationaljewish.org/conditions/tests-procedures/imaging/mri-intravenous-contrast-information
Radiological Society of North America, and American College of Radiology. (n.d.-a). MRI of the brain. RadiologyInfo.org. https://www.radiologyinfo.org/en/info/mri-brain
Radiological Society of North America, and American College of Radiology. (n.d.-b). Safety of MRI contrast materials. RadiologyInfo.org. https://www.radiologyinfo.org/en/info/safety-contrast
U.S. Food and Drug Administration. (2023, April 12). Information about gadolinium-based contrast agents. https://www.fda.gov/drugs/postmarket-drug-safety-information-patients-and-providers/information-gadolinium-based-contrast-agents
Footnotes
1. Magnetic Resonance Imaging (MRI) with contrast is the primary imaging mechanism used to identify glioblastoma in patients because it highlights abnormal tissue, including areas of tumor growth and necrosis.
2. Gadolinium, the contrast agent used in MRI, changes how hydrogen atoms in the body respond to the magnetic field, making abnormal areas “bright” on scans. It is generally safe, though mild side effects can include nausea or headache.
3. MRI does not involve radiation, unlike CT scans, making it a preferred method for repeated imaging in brain tumor patients.
4. A definitive diagnosis of glioblastoma requires a biopsy, even though MRI with contrast provides highly detailed images of the tumor.
5. Clinical presentation of glioblastoma can vary depending on tumor location and size. Common symptoms include headaches, seizures, neurological deficits, and increased intracranial pressure.
6. Survey methodology can be used to assess awareness of glioblastoma among students and professors, as well as gather data on patient experiences at medical centers such as UW Medicine (Alvord Brain Tumor Center) or Fred Hutchinson Cancer Center.